In polar covalent bonds the electrons are shared unequally as one atom exerts a stronger force of. Describe how electron placement determines chemical bonding stability.

Covalent Bond Ck 12 Foundation
Be sure to mention attractions between electrons and protons and the number of electrons in the outer energy level for the atoms in the final molecule.
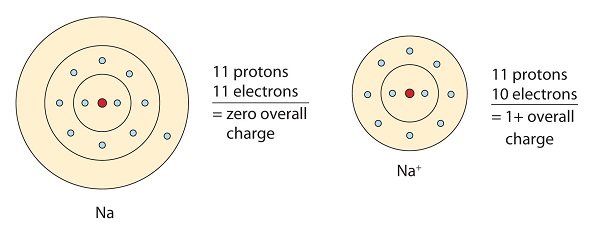
. Electrons shared in pure covalent bonds have an equal probability of being near each nucleus. A covalent bond is is sulphuric acid. Covalent bonding occurs between the atoms of non-metals.
However the nature of those atoms will determine if those electrons are shared equally or unequally. 423 Cl Cl Cl 2. They lie between the two nuclei of the bonding atoms.
One shares and the other trades. An atom that shares one or more of its. A type of chemical bond where two atoms are connected to each other by the sharing of two or more electrons.
Ionic And Covalent Bonding. As applied to covalent bonding what is meant by an unshared or lone pair of electrons. Covalent bonds form between two nonmetal atoms with identical or relatively close electronegativity values.
The electrons involved are in the outer shells of the atoms. Each electron is free to occupy either of the orbitals but both are more likely to be in the space between the nuclei of the bonded atoms. The shared electrons are typically near the middle of the bond between the 2 atoms in a covalent bond.
The outermost orbitals of the atoms overlap so that unpaired electrons in each of the bonding atoms can be shared. Describe the general location of the electrons in a covalent bond. They are going to be pulling the 2 nuclei together glue over lapping orbitals.
Jan 14 2015. A covalent bond may also be termed a molecular bond. Electrons shared in pure covalent bonds have an equal probability of being near each nucleus.
We refer to this as a pure covalent bond. A covalent bond in chemistry is a chemical link between two atoms or ions in which the electron pairs are shared between them. The shared electrons move in the orbitals around both atoms.
This type of covalent bond exists where the unequal sharing of electrons occurs due to the difference in the electronegativity of combining atoms. A covalent bond whose electron density is concentrated in the region directly between the nuclei. A region in space around the atoms nucleus where there is a probability of finding an electron.
ClCl Cl2 Cl. Reset E metals and nonmetals 1. Cl Cl Cl2 Cl Cl Cl 2.
We refer to this as a pure covalent bond. In the case of Cl 2 each atom starts off with seven valence electrons and each Cl shares one electron with the other forming one covalent bond. We refer to this as a pure covalent bond.
More electronegative atom will have a stronger pull for electrons. Covalent bonds form when electrons are shared between atoms and are attracted by the nuclei of both atoms. 7 rows The two main types of chemical bonds are ionic and covalent bonds.
9 rows The primary difference between ionic and covalent bonds is that an ionic bond is a. The electronegative difference between the atoms is greater than zero and less than 20. Describe the general location of the electrons in a covalent bond they occupy overlapping orbitals.
Electrons shared in pure covalent bonds have an equal probability of being near each nucleus. In a covalent bond electrons are shared between the two atoms that come together to form said bond. If 2 electrons pairs are shared 4 electrons are shared in all.
This type of bond may also be found in other chemical species such. Covalent Bonds Covalent bonds form when two atoms share electrons. A covalent bond forms when two non-metal atoms share a pair of electrons.
Describe the general location of the electrons in a covalent bond. They are in overlapping orbitals in the middle of two atoms. We refer to this as a pure covalent bond.
The greater the bond length in general the lower the average bond energy inversely Describe the general location of the electrons in a covalent bond. Briefly describe the process of covalent bonding between the carbon and the four hydrogen atoms to make a methane molecule. In polar covalent bonds the electrons are shared unequally as one atom exerts a stronger force of.
By overlapping orbitals the outer energy shells of all the bonding atoms are filled. Solution for Describe the location of electrons. Electrons shared in pure covalent bonds have an equal probability of being near each nucleus.
In pure covalent bonds the electrons are shared equally. Describe the octet rule in terms of noble-gas configurations and potential energy. General Chemistry - Standalone book MindTap Course List 11th Edition.
If the atoms that form a covalent bond are identical as in H 2 Cl 2 and other diatomic molecules then the electrons in the bond must be shared equallyWe refer to this as a pure covalent bondElectrons shared in pure covalent bonds have an equal probability of being near each nucleus. Two atoms with either equal or very similar electronegativity values the difference must not exceed 05 come together to form a nonpolar. In the case of Cl 2 each atom starts off with seven valence electrons and each Cl shares one electron with the other forming one covalent bond.
Covalent bonds form when electrons are shared between atoms and are attracted by the nuclei of both atoms. The general location of electrons in a covalent bond is that electrons are shared in pairs between 2 atoms. In the case of Cl 2 each atom starts off with seven valence electrons and each Cl shares one electron with the other forming one covalent bond.
In pure covalent bonds the electrons are shared equally. In the case of Cl 2 each atom starts off with seven valence electrons and each Cl shares one electron with the other forming one covalent bond.

12 2 Ionic And Covalent Bonds Chemistry Libretexts

Covalent Bond Definition Properties Examples Facts Britannica

0 Comments